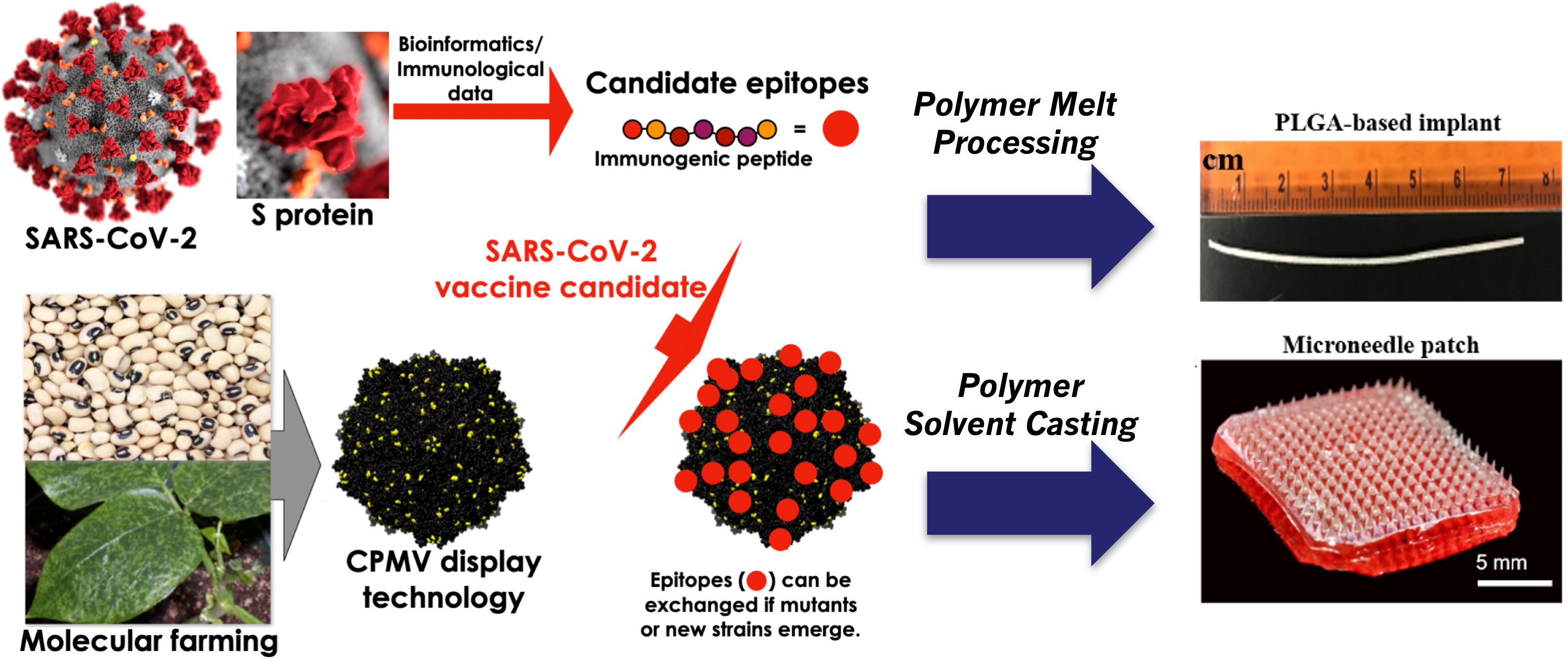
 The COVID-19 pandemic highlights the need for platform technologies enabling rapid development of vaccines for emerging viral diseases. The current vaccines target the SARS-CoV-2 spike (S) protein and thus far have shown tremendous efficacy. However, the need for cold-chain distribution, a prime-boost administration schedule, and the emergence of variants of concern (VOCs) call for diligence in novel SARS-CoV-2 vaccine approaches. We studied 13 peptide epitopes from SARS-CoV-2 and identified three neutralizing epitopes that are highly conserved among the VOCs. Monovalent and trivalent COVID-19 vaccine candidates were formulated by chemical conjugation of the peptide epitopes to cowpea mosaic virus (CPMV) nanoparticles and virus-like particles (VLPs) derived from bacteriophage Qβ. Efficacy of this approach was validated first using soluble vaccine candidates as solo or trivalent mixtures and subcutaneous prime-boost injection. The high thermal stability of our vaccine candidates allowed for formulation into single-dose injectable slow-release polymer implants, manufactured by melt extrusion, as well as microneedle (MN) patches, obtained through casting into micromolds, for prime-boost self-administration. Immunization of mice yielded high titers of antibodies against the target epitope and S protein, and data confirms that antibodies block receptor binding and neutralize SARS-CoV and SARS-CoV-2 against infection of human cells. We present a nanotechnology vaccine platform that is stable outside the cold-chain and can be formulated into delivery devices enabling single administration or self-administration. CPMV or Qβ VLPs could be stockpiled, and epitopes exchanged to target new mutants or emergent diseases as the need arises.
The COVID-19 pandemic highlights the need for platform technologies enabling rapid development of vaccines for emerging viral diseases. The current vaccines target the SARS-CoV-2 spike (S) protein and thus far have shown tremendous efficacy. However, the need for cold-chain distribution, a prime-boost administration schedule, and the emergence of variants of concern (VOCs) call for diligence in novel SARS-CoV-2 vaccine approaches. We studied 13 peptide epitopes from SARS-CoV-2 and identified three neutralizing epitopes that are highly conserved among the VOCs. Monovalent and trivalent COVID-19 vaccine candidates were formulated by chemical conjugation of the peptide epitopes to cowpea mosaic virus (CPMV) nanoparticles and virus-like particles (VLPs) derived from bacteriophage Qβ. Efficacy of this approach was validated first using soluble vaccine candidates as solo or trivalent mixtures and subcutaneous prime-boost injection. The high thermal stability of our vaccine candidates allowed for formulation into single-dose injectable slow-release polymer implants, manufactured by melt extrusion, as well as microneedle (MN) patches, obtained through casting into micromolds, for prime-boost self-administration. Immunization of mice yielded high titers of antibodies against the target epitope and S protein, and data confirms that antibodies block receptor binding and neutralize SARS-CoV and SARS-CoV-2 against infection of human cells. We present a nanotechnology vaccine platform that is stable outside the cold-chain and can be formulated into delivery devices enabling single administration or self-administration. CPMV or Qβ VLPs could be stockpiled, and epitopes exchanged to target new mutants or emergent diseases as the need arises.